The US ship, the MV Cape Ray, was fitted out with two $5m (£3m)
mobile hydrolysis systems, developed in 2013 by US military
researcher, specifically to deal with the chemicals.
SARIN = Isopropyl-Methyl-Phosphoryl-Fluoride
SYRIAN CONFLICT:- All Sarin Gas Chemicals Destroyed …
The MV Cape Ray was fitted out with mobile hydrolysis
system specifically to deal with the chemicals. See below ♦
Continue reading the main story:
The international chemical weapons watchdog says that all the precursor chemicals
for sarin gas removed from Syria have been destroyed.
The 580 tonnes of chemicals were neutralised on board a specially built American ship in the Mediterranean Sea.
The Organisation for the Prohibition of Chemical Weapons (OPCW) said the ship’s crew have now begun to neutralise 22 tonnes of sulphur mustard.
The process is part of a Russian-US deal to eliminate Syria’s arsenal.
The deal was drawn up after hundreds of people died in a sarin attack in the Ghouta area outside Damascus on 21 August last year.
The US ship, the MV Cape Ray, was fitted out with two $5m (£3m) mobile hydrolysis systems, developed in 2013 by US military researcher, specifically to deal with the chemicals.
A UN team was sent into Syria to oversee the removal of President Assad’s chemical stockpile OPCW chief ambassador Ahmet Uzumcu announced the completion of this stage of the process on Wednesday.
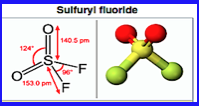
The two hydrolysis systems neutralised the precursor chemicals – methylphosphonyl difluoride – by mixing them with fresh water and “reagents” and then heating the mixture to reduce its toxicity.
Once the ship has processed the remaining sulphur mustard chemicals, all of the resulting effluents will be transported to Finland and Germany for disposal at land-based facilities.
 ♦
♦
…This means the 280 grams of sarin could be manufactured for
$130 and could kill over 10,000 people.
Sarin is also known as GB.
BUY THE INGREDIENTS → HERE ← Cheap!
• dimethyl methylphosphonate
• phosphorus trichloride
• sodium fluoride
• alcohol