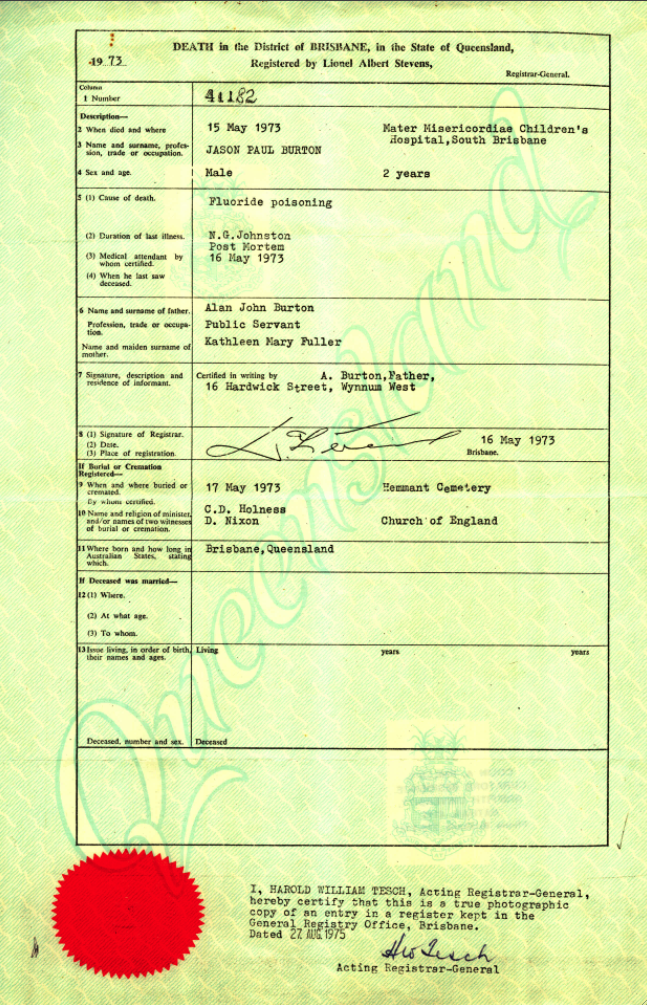
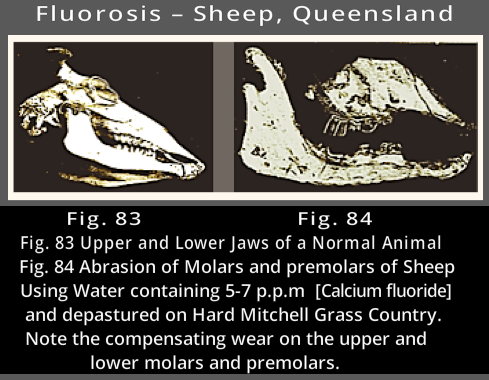
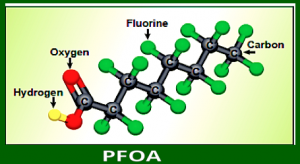
1939
|
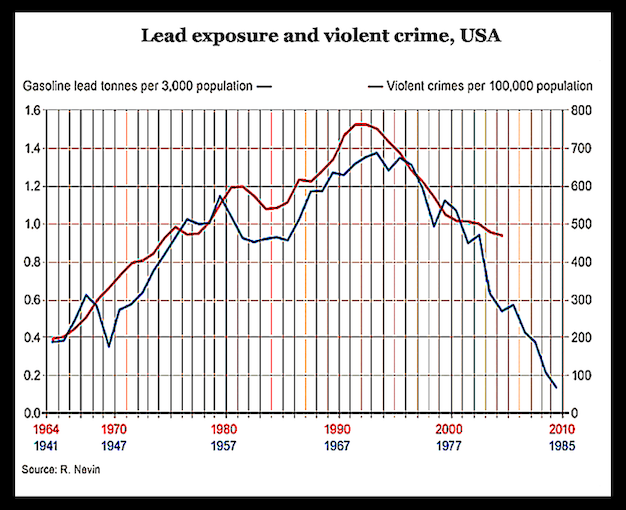
The concept of fluoridation arises as an alternative method of disposing of industrial waste chemicals, where disposal expense is replaced by profit. This fact was confirmed with approval in a 1983 letter written by Rebecca Hanmer, Assistant Administrator, from EPA Office of Water.
|
1939
|
The Hatch Act was passed after revelations that employees of the WPA, a New Deal agency, were pressured to make political contributions. The new Act protected against a politicized federal work force. It also prohibited any federally funded agency, whether county, state, or federal, from trying to influence public referenda. Since the beginning of the effort to fluoridate water in the 1940s, however, the Hatch Act has been repeatedly and flagrantly violated
|
1939
|
On Sept 29, Mellon Institute scientist, Gerald J. Cox, begins his major role in the promotion of fluoridation by saying, “the present trend toward removal of fluorides from food and water may need reversal”.Note: Scientist Cox also had this to say in 1939: “Fluorides are among the most toxic of substances. Mottled enamel results from as little as 0.0001 percent of fluorine in the drinking water. Every use of water must be examined before fluoridation can begin”. (Journal of the American Water Works Assn. pp. 1926-1930, Nov 1939). Despite all of this, Alcoa sponsored biochemist, Gerald J. Cox, fluoridates rats in his lab and mysteriously concludes that “fluoride reduces cavities”. He makes a public proposal that the US should fluoridate its water supply. Cox begins to tour the US, stumping for fluoridation.
|
1939
|
The American Water Works Association decided there was sufficient evidence about fluoride to classify it as a hazardous material, like lead and arsenic. It then suggested that drinking water should contain no more than 0.1 ppm fluoride.
|
1941
|
Instead of forbidding the dumping of fluoride in water, the USPHS regulations set 1.0 ppm of fluoride as the maximum tolerance allowed in a public water supply. This allowed industries to continue to dump fluoride wastes into rivers.
|
1941
|
In December, Japan attacks Pearl Harbor. All anti-pollution regulations are suspended. Many parts of America now suffer hydrogen fluoride air pollution on an unprecedented scale. Major fluoride hazards develop in war materials production of WWII, consolidating government collusion with big industry on a coverup of fluoride hazards.
|
1942
|
In England, a Lancet report showed that out of 589 London children, 28% had mottled teeth. According to Alcoa’s chief chemist and the USPHS, London’s drinking water should contain well over one ppm fluoride to account for this. Tests showed just 0.19 ppm. Clearly, hydrogen fluoride was the cause. In this case, it was surely related to the heavy use of coal for fuel, a known source of HF.
|
1942
|
Hydrogen fluoride supplants sulfuric acid as a catalyst in the production of high test gasoline in Los Angeles. One such plant required 500-750 tons of HF yearly (Fluorine Industry Chem. and Met. Eng., 52:94-99 Mar. 1945).
|
1943
|
Planning began on the Newburgh, NY, Fluoridation Demonstration Project. Atomic bomb program scientists played a prominent but unpublicized role in this first US fluoridation experiment. Fluoride was the key chemical in atomic bomb production. Millions of tons of fluoride were needed for the manufacture of bomb-grade uranium and plutonium for nuclear weapons. Today, memos released under the Freedom of Information Act show that scientists from the atomic bomb program secretly shaped and guided the Newburgh fluoridation experiment. This reveals the US government conflict of interest and its motive to prove fluoride safe.
|
1944
|
Oscar Ewing is put on the payroll of the Aluminum Company of America as an attorney, at an annual salary of $750,000.
|
1945
|
Program “F” is implemented by the US Atomic Energy Commission (AEC). This is the most extensive US study of the health effects of fluoride – a key chemical component in atomic bomb production. One of the most toxic chemicals known to man, fluoride was found to have marked adverse effects to the central nervous system. But much of the information was classified “secret” in the name of national security because of fear that lawsuits would undermine full-scale production of atomic bombs.
|
1946
|
With no new evidence of safety, and no stated reason, USPHS raised the maximum tolerance level of fluoride in public water supplies to 1.5 ppm.
|
1947
|
Alcoa lawyer, Oscar Ewing, is appointed head of the Federal Security Agency, later HEW, a position that places him in charge of the USPHS. He is the second Alcoa executive (after Andrew Mellon) to direct the course of the Public Health Service, completing its mutation into a virtual pawn of big industry. Under Ewing, a national fluoridation campaign rapidly materializes, spearheaded by the USPHS. Over the next three years, eighty-seven cities were fluoridated. This included the control city of Muskegan in the original Michigan experiment, thus wiping out the most scientifically objective test of safety before the test was half over.Ewing’s public relations strategist was Edward L. Bernays, Sigmund Freud’s nephew, who pioneered Freudian theory toward advertising and government propaganda (see Bernays’ 1928 book, Propaganda). Because of Bernays, people would be induced to forget that fluorides were toxic poisons. Opponents to the fluoridation program were painted as deranged. In 1996 they would be painted as civil rights activists, crackpots, and right-wing loonies. As the newspapers were heavily influenced by industry advertisers, they became key dispensers of such propaganda.
|
1948
|
The Donora Death Fog was the second major air pollution disaster in history. It was caused by the accumulation of stagnant hydrogen fluoride gas from steel and zinc smelters in a narrow industrialized valley. Six thousand of the 13,000 residents of this Pennsylvania town’s population became ill, and on the fourth day seventeen died. A leading forensic chemist, Philip Sadtler, investigated the tragedy and reported strong evidence of acute fluoride poisoning. His report appeared in Chemical and Engineering News under the headline, FLUORINE GASES IN ATMOSPHERE AS INDUSTRIAL WASTES BLAMED FOR DEATH AND CHRONIC POISONING OF DONORA AND WEBSTER. The USPHS whitewashed the incident in their report (see Public Health Bull. No. 306, Washington, D.C., 1949). Their conclusion was: No pollutant present could have caused the disaster. The following are excerpts from a critique of that report by Frederick B. Exner, MD:”A 173-page report tells us that there had been no unusual kind or amount of pollution, and that no pollution present could have caused the trouble. Sampling methods of doubtful reliability were applied at arbitrarily selected times and places, and the results averaged with no attempt at proper weighting. Calculations therefrom, replete with arithmetical errors and discrepancies, were combined with outright guesses to arrive at estimates of emission.They guess that 210 tons of coal burned in homes emit 30 lb. of fluorine but that 213 tons burned in the blooming-mill boilers emit only four lb. No possible reason for the difference is offered.On page 104, waste gas from the blast furnace contains 4.6 mg of fluorine per cubic meter. On page 108 it contains one-tenth as much.Calculations for open-hearth emission show a discrepancy of several thousand fold, with no way to know where the error lies.The biological studies and general air sampling are similarly inappropriate and meaningless. Air samples at twelve arbitrarily selected points between Feb. 16, and April 27, 1949, can tell us nothing about concentrations during the episode.”Test results of a study made of the Donora disaster by US Steel have been withheld from public view to this day. This is unmistakable evidence of an effort to cover up highly toxic HF emissions.
|
1948
|
As a direct consequence of the Donora disaster, USPHS began quietly sampling fluorides in the air over 27 major cities across the country. This sampling turned up serious HF air pollution (up to 80 ppb) in the following twelve cities: Pittsburgh, Baltimore, Chicago, Cleveland, Milwaukee, St. Louis, Philadelphia, San Francisco, Buffalo, Denver, Oklahoma City, and Indianapolis (see Register of Air Pollution Analyses, US Department of Health, Education and Welfare. USPHS, Washington DC, 1949-1961).
|
1950
|
The new hydrogen fluoride air pollution data collected by the USPHS presented a major problem. Data gathered showed HF contamination up to 80 ppb, more than ten times what had been proposed for standards.Strong circumstantial evidence suggests that the camouflage strategy adopted more than a decade earlier by Alcoa in Pittsburgh was to influence the strategy adopted by the USPHS: If the nation’s twelve cities with the most serious HF air pollution were fluoridated, this expensive-to-correct problem would be camouflaged. Dental fluorosis could then be attributed to the water, and authorities could describe mottled teeth as an “acceptable trade-off” for the claimed caries preventing properties of fluoridated water. To bring this about, the Great Fluoridation Experiment underway in Grand Rapids and three other cities was declared a success in June 1950, five years before the experiment would be complete. Before a single tooth had fully developed under the influence of the experimental fluoridated water, USPHS claimed a reduction in tooth decay of between 50 and 60 percent. (Dean, H. T. et al., Studies on Mass Control of Dental Caries through Fluoridation of the Public Water Supply, Public Health Report 65, 1950).This “success” then allowed USPHS to rush out to fluoridate the twelve cities with major HF air pollution and thereby camouflage the toxic air problems. All twelve cities were fluoridated in the following five years. The same camouflage was to be carried out two years later by Alcoa in Australia.
|
1950
|
Two years after the disaster in Donora, when the USPHS found serious HF air pollution across the country, their analytical method was changed from measuring the level of HF to measuring the level of fluoride ions in the air. Deception clearly motivated this change. Fluoride ions, like fluorine gas, are relatively rare toxic air emissions. By pretending that fluoride ions, not the far more harmful HF, was the concern in contaminated air, the USPHS avoided exposure of incriminating HF data which it thereby managed, once again, to ignore.
|
1950
|
From 1950 to 1951, Alcoa advertises sodium fluoride for addition to water supplies.
|
1950
|
The Journal of the American Dental Association, (30:447, 1950), features an article by Dr. G. J. Cox, University of Pittsburgh, who says, “To solve the esthetic problem for victims of mottled enamel, porcelain facings, jacket crowns, or even dentures may be required”. Note: The public is expected to bear the cost of what is being done to them while the dental industry profits.
|
1951
|
Early in 1951 Oscar Ewing allocated $2 million to “promote fluoridation nationwide”.
|
1951
|
Oscar Ewing was sponsoring a bill which the conservative American Medical Association claimed would be the first step toward socialized medicine. The AMA appealed to its members for a “fighting fund” to defeat the Bill and $3 million was raised. But at the AMA convention in Los Angeles, Ewing notified the committee that the bill was to be withdrawn. That same committee, which had never before considered the subject, suddenly released a statement saying that the AMA totally endorsed the “safety of fluoridation”. At that time there was not one published paper providing evidence to support the AMA endorsement. But from then on, the AMA left fluoridation to dentists – and to those powerful forces which were manipulating the dental trade association (ADA).
|
1952
|
The ADA Journal instructs its dentists not to discuss their personal opinions about fluoride. Here is blatant evidence of ADA political bias.
|
1952
|
In London, the greatest toxic fog disaster in history occurred from December 5-9 in a temperature inversion. Hydrogen fluoride (HF) gas was the culprit, as in the two earlier major disasters. During those five days there were 2,000 excess deaths in London, and some 10,000 more people were wiped out in the surrounding Thames Valley. Similar episodes, both before and after this one, occurred in London. In 1945, a noxious fog brought death to 600; in 1956, to 500; and in 1957, to 400 (Air Pollution, published on behalf of the World Health Organization, Columbia University Press, N.Y., 1961, p. 175).Shocking as it is, the toll of lives does not tell the whole story. Neither the assessments of the toxic air disasters, nor tests establishing maximum contaminant levels, take into account the widespread effects on mental function brought about by HF poisoning. Human behavior is exquisitely sensitive to minute traces of hydrogen fluoride – in the parts per billion range. In London, it is likely that millions were so affected. This includes symptoms of confusion, fatigue, partial loss of memory, and mental dullness and apathy. The condition identified in 1982 as chronic fatigue syndrome is currently of undetermined origin, and is now increasingly widespread. The same symptoms are caused by HF air pollution. Research on hydrogen fluoride is lacking, and funding is not available.
|
1952
|
USPHS officials, Drs. Dean, Arnold and McClure, concentrate their efforts to introduce fluoridation into Australia and New Zealand, providing more evidence for an underlying industrial motivation.
|
1952
|
Alcoa starts construction of the first aluminum smelter in Australia, two miles from the small town of Beaconsfield, Tasmania. The following year, Beaconsfield became the first town in all of Australia to install water fluoridation. Dental fluorosis could then be attributed to the water as an “acceptable trade-off” for prevention of caries (unproven). Beyond coincidence, here is more evidence of the industrial strategy to camouflaging airborne HF poisoning by fluoridating the water supply.
|
1955
|
The Kettering Laboratory in Cincinnati has become the largest organization of its kind in the world with a staff numbering about 120. Its specified purpose is to investigate chemical hazards that develop in American industrial operations (to prevent a replay of the litigation that plagued European industry and gave American industry a competitive edge).
|
1956
|
On Jan 26, Procter & Gamble ran a full page ad in the New York Times, proclaiming Crest toothpaste “an important milestone in medicine”, comparing it to Dr. Fleming’s discovery of penicillin. P & G published no evidence supporting their extravagant claims. Harold Hillenbrand, secretary of ADA responded saying there was no evidence that any fluoride paste could prevent tooth decay. Initially there was an FDA warning label on Crest, but it disappeared in 1958, without explanation, and did not reappear until nearly forty years later.
|
1957
|
Alcoa announces the direct sale of sodium fluoride to cities and towns – for fluoridation of drinking water. A decade later, when it was found that phosphate fertilizer companies could sell fluorides from their smokestack scrubbers for even less money, Alcoa was priced out of the fluoride dumping market.
|
1957
|
The American Dental Association receives $6,453,816 in federal funds, from 1957-1973.
|
1958
|
The World Health Organization (WHO) establishes an Expert Committee in Geneva to study fluoridation. At least five of the seven committee members had promoted fluoridation in their own countries. The American proponent, Professor H. C. Hodge, had some of his research financed by the Atomic Energy Commission, which was confronted with serious fluoride disposal problems from uranium processing. Professor Ericsson, the member from Sweden and a prominent advocate of fluoridation in Europe, was the recipient of a USPHS grant and received royalties from Sweden’s toothpaste industry. Such are the sources of the WHO endorsement of fluoridation.
|
1959
|
Reynolds Metals Co. built an aluminum smelter on the Gulf of St. Lawrence, upwind of a Mohawk Indian Reservation. Fifteen-hundred Mohawk Indians farmed on their island Reservation. Forty-five farmers had forty cattle barns and 364 dairy cattle. Cattle became lame and many cows died. In 1977, there were just 177 left. The farmers themselves were found to have muscular and skeletal abnormalities. The Mohawk way of life became the victim of a preventable man-made plague caused by hydrogen fluoride.
|