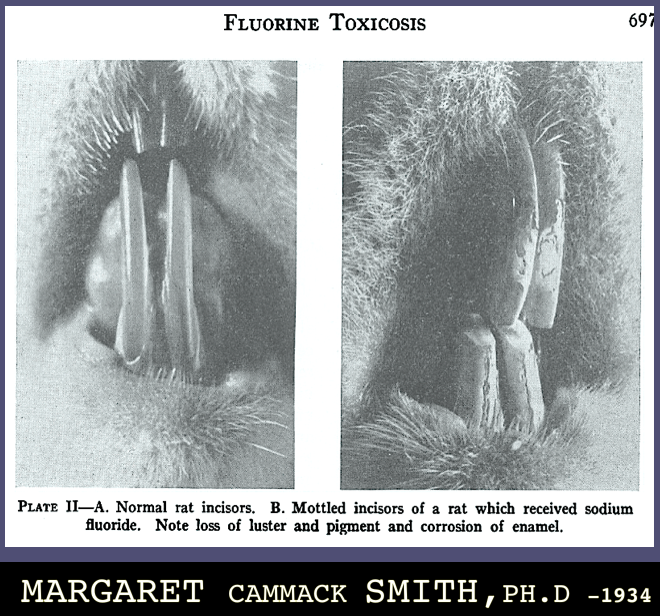
“Both the very young and the very old are
most likely to be adversely affected
after exposure to fluorides.”

Original text → HERE ← Thank you Robert
Robert L. Isaacson Distinguished Professor of Psychology Binghamton UniversityA note on terminology: Fluorine is an element in the halogen group as are chlorine and iodine. Of all the known elements, fluorine is the most chemically reactive, most powerful oxidizing, and most electronegative element. It is a stronger oxidizing element than ozone. It reacts with many compounds at room temperature. It is never found in its pure form in nature.Fluoride: Any combination of fluorine with another element or chemical group of elements. Thus, the addition of fluorides to the drinking water can indicate the addition of a large number of chemical agents. The most commonly used fluorides for this purpose are sodium fluoride, NaF, and compounds that contain both fluorine and silicon. Such agents are collectively called “Fluorosilicates.” They include fluorosilicic acid, fluorosilicate, hydrofluosilicic acid, and hexafluorosilicic acid.In 2003 when I accepted an invitation to join the National Research Council’s Committee formed to evaluate the EPA standards for the amount of fluoride that should be allowed in our drinking water, I had no fixed opinion on whether or not fluoride should be added to drinking water. Probably I was asked to serve on the committee because I had organized a series of experiments published between 1993 and 1998 using rats to study the effects of chronic administration of aluminum fluoride in their drinking water. My primary interest was in the effects of aluminum on the brain and behavior. Aluminum fluoride was used because fluoride facilitates the passage of aluminum into the brain. At the time, aluminum was considered by a number of scientists to be an important factor in Alzheimer’s disease. Scientists are still actively investigating this possibility. Our studies had to include the investigation of the effects of the fluoride since the aluminum and the fluoride readily become associated after ingestion. In essence we wanted to know the effects of the aluminum, fluoride, and the aluminum- fluoride complex. 1
1
In my more than three years working on the National Research Council Committee I learned about the many influences fluoride has on the nervous system and the brain. I also learned about the variety of ways in which people become exposed to it and the work that had been done in trying to determine if fluorides were a hazard to human health and well being. The results and recommendations of this Committee were published late in 2006.2 Slowly, I came to the conclusion that there were strong experimental and clinical indications that fluorides present health hazards to people in many ways. The more I learned, the more I became convinced that the addition of fluorides to drinking water was, and is, a mistake. Accordingly, I decided to share some of my conclusions with any who might wish to know them. Fluorine-containing compounds can affect every living animal and person. Exposure to fluorides can come from the air, the water, and the foods we eat. Fluoride compounds were long used as insecticides. They were especially effective for ants and roaches. Their containers were always boldly marked as a poison and there were warnings on the label to keep them well away from children. This is mentioned only to note that for many years fluorides have been considered to be major health hazards. In regard to health the total accumulation of fluorine in the body is important. Only about half of the amount of fluorides taken in by a person is excreted. The rest stays in the body. Toxic effects are determined by the amount of fluoride stored in the body, current exposure level, and age at the time of exposure. In addition each person has his or her own tolerance level for fluorides. Once this level is exceeded however, dysfunctions of body and/or brain will occur. How these dysfunctions will be expressed depends on the genetic makeup and past experiences of the person. Another factor
that helps determine a person’s sensitivity to fluoride is their age. Both the very young and the very old are most likely to be adversely affected after exposure to fluorides. As noted, different people exhibit a wide range of toxic reactions to fluorides. Some people affected by fluorides complain of general weakness and chronic fatigue, others complain of cramp-like pains in the abdomen, or nausea. Still others express toxin-induced effects by diminished vision, headaches, migraine attacks, or pains in muscles and joints. These fluoride effects have been described in books by Leo Spira (1950, 1959)3 and George Waldbott and his associates (1978).4 It is difficult to determine whether or not a given set of symptoms is a consequence of fluoride intake. It is first necessary to rule out the presence of other diseases that could produce the observed symptoms. A correct diagnosis is best shown by repeated observations of an individual when drinking pure water or water contaminated with a fluoride. These exposures must last for periods of a week or two under conditions in which the patient doesn’t know which type of water is being consumed. If the symptoms disappear when the person is drinking pure water and return with the resumption of drinking the fluoride- treated water, this is evidence that the problems arise from the fluoride. Leo Spira and George Waldbott and his associates used this type of experimental approach in their research. Since people vary so much in their sensitivities to fluorides and also in the nature of their symptoms caused by this toxin, determination of a uniform “safe” level of exposure for everyone is impossible. In a way, fluorides are like ozone: there is no really “safe” level that would protect everyone. The Congressional Safe Drinking Water Act instructed that the level of fluoride in drinking water should be set so as to be safe for everyone
regardless of age or overall health. Increasing the problems that can be induced by fluorine in its different forms is its ability to enhance the effects of other toxins to which we are exposed. For example, fluorides in the drinking water accelerate the absorption of lead, aluminum, and silicon into the body and brain. The toxic effects of lead have been known for hundreds of years. In recent years the focus of attention has been on the learning deficits lead produces in children. The mechanisms proposed for the induction of this effect are not known entirely but there is evidence that many of the most important neurotransmitters of the brain are being affected. These include alterations in dopaminergic, cholinergic, and glutaminergic systems as well as in the “supportive” glia cells of the brain. There is also evidence that lead toxicity may go beyond impairments of intelligence. Indeed, lead toxicity may produce behavioral changes that include loss of impulse control and a related increase in the frequency of violent acts.5 The health hazards associated with enhanced incorporation of lead are not induced by all fluorides but primarily, and maybe only, by the addition of a silicofluoride to our drinking water. The fluoride most often added to our drinking water is hexafluorosilicic acid. This fluorosilicate dissociates when it enters the body. One component contains silicon and another fluorides. As a consequence when silicofluorides are added to our drinking water there are really two toxic hazards: one coming from the fluoride and another from the silicon. Silicon can produce its own toxic effects including the formation of solids (silica and silicates) that can lodge anywhere in the body. In addition the silicon portion also can generate destructive hydroxyl ions in many organs including the brain. The brain damage caused by the production of these free radicals has been related to anti-social behavioral
actions and violence.6 Recently data from 327 towns and cities, some having fluoridated water and others not, have been compared in terms of crime rates. All the communities with fluoridated water had substantially higher rates than did those with non-fluoridated water. This indicates that fluorides can act to enhance the damage being done by other toxins. The impairment of intelligence from lead toxicity is now well established. It is possible that fluorides can produce negative effects on measured intelligence also. The country devoting the greatest attention to this possibility is China. As of February 2007, several groups of Chinese investigators had published over 20 scientific papers on this topic. Scientists from many different areas of China participated in these investigations. The children studied in these reports ranged in age from 4 to 14. All were tested by the same or very similar standardized I.Q. tests. Overall the results came from children tested at different places, at different ages, and tested by different investigators. All the results from China have found that communities with high levels of fluoride in their drinking water have fewer children scoring at the “bright” end of the intelligence spectrum than communities with low or no level of fluoride. Since China does not fluoridate their drinking water, the Chinese studies compare the I.Q. scores of children from towns and school areas that differ in the amount of fluoride naturally present in their water supplies. While not all of Chinese studies were perfectly designed, the large number of studies showing the same pattern of results calls for our attention. A negative effect of fluoride on intelligence seems to be a possibility. Other studies in China have indicated that fluoride exposure in the drinking water of mothers during the 6th to 8th months of pregnancy can
produce anatomical changes in the fetal brains. There are also reports of impaired responsiveness to visual and auditory stimuli in babies in the first three days after birth induced by the intake of fluoridated water by young mothers during gestation.7 The ingestion of fluoride tends to increase the uptake of aluminum by the brain. In the studies done in my laboratory the increase in aluminum in the brains of rats was not a function of the amount of aluminum fluoride given the animals in their drinking water. The smallest dose of aluminum fluoride produced about the same amount of aluminum in the brain as a dose 10 or even a 100 times larger. A small amount of fluoride seems capable of opening aluminum pathways to a maximal degree. It is of great interest that the relative risk of having Alzheimer’s disease is increased when individuals had high amounts of aluminum in the brain coupled with low amounts of fluoride.8 Another observation of interest is that aluminum by itself may not exert toxic affects on the nervous system. It may only become a toxin after joined together with a fluoride to become an aluminum fluoride. 9 The chronic administration of fluorides in rats produces changes in the microscopic structure of the brain. There were significant losses of cells in areas of the hippocampus and the neocortex. Many apparently dead or dying cells were found in areas analogous to locations in which similar dying cells are found in the brains of Alzheimer’s patients. A common and, perhaps universal, characteristic of dementia is a reduction of aerobic metabolism in the brain. The blood supply reaching the brain is the primary supplier of oxygen and nutrients. Reductions in this sole source of brain energy can be due to a number of physical or chemical changes. When the brains of animals chronically exposed to aluminum fluoride were examined histologically, deposits of aluminum-based crystals
were found along the walls of both large and small blood vessels in the brain. Similar deposits were also found in the center of many vessels suspended by collagen fibers. These deposits decreased the normal transfer of oxygen from the red blood cells to the brain since they must have created turbulence in its blood flow. It is of historical interest that Alois Alzheimer, the man for whom a type of dementia was named, noted that most patients with this disorder suffered from atherosclerosis in addition to other brain anomalies. This condition is one in which there are deposits formed on the sides and in the center of arteries in the brain. The deposits disrupt the flow of blood to the brain often cause severe brain damage. Brain functions are entirely dependent on the availability of oxygen. The brain itself consumes 20% of all the oxygen used by the entire body. The brain area most affected by the reduction in oxygen availability is the forebrain. The lower centers of the brain, namely the midbrain and hindbrain, are more resistant to oxygen deprivation. This is why the higher functions of the brain are the first to be affected, as well as the most affected, by oxygen deprivation. Basic motor and visceral functions are often spared even in patients with profound interruptions of normal blood supplies to the brain. One of the best-known chemical alterations produced by fluorides is a reduction in cholinesterases, including acetylcholinesterase. Fluorides also directly affect the actions of many of other important neurotransmitters in the brain. Fluorides seem to have a special attraction to acetylcholine. Nerve cells that synthesize this transmitter have numerous projections to many forebrain areas, including the neocortex and deeper areas of the brain that provide information to the neocortex. Not only do fluorides change the amount of the acetylcholine in the
7
brain, they selectively block certain receptors that respond to this transmitter. Fluoride reduces the number of one type of “nicotinic receptors” for acetylcholine. Some other nicotinic subtypes are not affected.10 Added to all of the other alterations in structure and function of the brain caused by fluorides, the opportunity for mental and behavioral changes are almost limitless. While the cholinergic system of the brain has been most studied in regard to the effects of fluoride, it is not the only neural transmitter affected. It is likely that all neural transmitter systems are affected by fluoride intake, directly or indirectly. Other anomalies related to fluoride intake are found in many other chemical systems of the brain. During the period from 1956 to 1963, the endocrinologist, Ionel Rapaport, presented evidence of a link between fluoride exposure and the numbers of babies born with Down’s syndrome, (Trisomy 21). For a number of years the only follow up to his work was in the form of epidemiological comparisons between the number of births of such children both to mothers living in fluoridated drinking water vs. the number of such born to mothers births in or non-fluoridated drinking water areas. The demographics of the two or more areas being compared were not fully taken into account in most of the studies. Maternal ages were also not taken into consideration. Overall, the “follow up” studies to Rapaport’s report were not decisive but none of them failed to rule out his original findings. Furthermore, a determination of fluoride effects using standard epidemiological procedures cannot provide convincing information. This is because it is impossible to find populations virtually the same in all regards except for the amount of fluoride in their drinking water. Another problem arises from the difficulty in accurately determining the number of Down’s
syndrome children born. Some investigators use the number of birth certificates on which the attending physician notes that the baby had Down’s syndrome. Other investigators use only closed hospital records made sometime later. Still other investigators use both. Neither method is perfect. The use of entries on hospital records would seem to be the most accurate method since physicians seldom enter the nature of possible deformities like Down’s syndrome on birth certificates after delivery. Indeed because of the possibility of making a mistake from delivery, the diagnosis is not often made until a determination can be made by laboratory results. Probably the best collection of relevant data comes from a study of births of children born in two areas of Atlanta, Georgia, as reported by Erickson et al. in 1976. Two different estimates of the number of Down’s children and normal children were presented. One estimate of Down’s syndrome births was made by the examination of copies of birth certificates and the other was based on hospital records. A re-examination of Erickson’s data by Burgstahler11 showed an overall enhancement of Down’s syndrome births to mothers from the fluoridated area. Later, in 1998 Takahashi did a fine grain analysis of data from a number of sources that included the corrected numbers from the 1966 Erickson report.12 In the Takahashi report a clear-cut relationship between fluoride exposure and the number of affected children was found in mothers 30 years of age and younger. Recently, Juan C. Molino13 and I using only data from hospital records found the same age-fluoride-Down’s syndrome birth effect. In his report Takahashi extended the analysis of his data through the use of a regression analysis. He wanted to determine if there could be any dose that would not increase the likelihood of having a Down’s syndrome child. According to his calculations there was no such dose. All doses of
fluoride caused some enhancement of the likelihood of a woman having such a child. There are other data supporting the idea that fluorides can induce genetic alterations. Evidence indicating biochemical interactions of fluoride with the genetic mechanisms of cell division are presented in the NRC report on Fluoride in the Drinking Water. (See Endnote 2) When the possible benefits and possible dangers of fluoride are considered there really is no comparison. Consider the following: There is no known benefit of adding any form of fluoride to our drinking water. Who would want to increase chances of having a less than perfect child? Who would wish to take a chance on a possible reduction of their own mental capacity? Who would want to have their personality altered by fluoride induced alterations in their brain chemistry? Who would want to increase their odds of developing Alzheimer’s disease? Eliminating the addition of fluoride to our drinking water would remove these possibilities. The cost of doing this is zero. In fact it would enrich the communities now adding fluorides to their drinking water.
1.
Endnotes Varner, J. A., Huie, C. W., Horvath, , W. J., Jensen, K. F., and Isaacson, R. L. (1993) Chronic AlF3 administration: II. Selected histological observations. Neurosci. Res. Comm. 13:99-104.Varner, J. A., Jensen, K. F., Horvath, W. J. and Isaacson R. L. (1998) Chronic administration of aluminum fluoride or sodium fluoride to rats in drinking water: alterations in neuronal and cerebrovascular integrity. Brain Res., 784: 284-298.Varner, J. A., Horvath, W. J., Huie, C. W., Naslund, H. R., and Isaacson, R. L. (1994) Chronic aluminum fluoride administration. Behav. Neural Biol., 61: 233-241.Isaacson, R. L., Varner, J. A., and Jensen, K. F. Toxin-induced blood vessel inclusions caused by the chronic administrations of aluminum and sodium fluoride. Ann. NY Acad. Sci., 825:152- 166. The final report of the committee was published by the National Academies Press in December 2006, entitled “Fluoride in drinking water.” It can by obtained from the National Academies Press and by special order from any bookstore. The electronic link to the NRC/NAS publication sites:
2.
1 – 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13.http://nap.edu/catalog/11571.html. Spira, L. The drama of fluorine, archenemy, of mankind. Milwaukee: The Lee Foundation for Nutritional Research, 1950, 1959. Waldbott, G. L. Fluoridation the great dilemma, Lawrence, KA: Coronado Press, 1978. Masters R. D., Coplan, M.J. Association of silicofluoride treated water lead with elevated blood lead. Neurotoxicology, 2000. 21:1091-1100. Masters, R. D., Coplan M. J. A dynamic, multifactoral model of alcohol, drug abuse and crime: Linking neuroscience and behavior to toxicology. Soc. Sci. Information, 1999, 38: 591-624. Seavy, J., (2005) Water fluoridation and crime in America. Fluoride, 38:11-22. Du Li. (1992) The effect of fluorine on developing human brain. Chinese Journal of Pathology, 21:218-20. Li Jing, Yao L., Shao, Q-L, and Wu, C-Y. (2004) Effects of high fluoride level on neonatal neurobehavioral development. Chinese Journal of Endocrinology, 23: No.5. Belovjovic, G., Jakovlevic, B. (1999) Aluminum and Alzheimer’s disease. Spr. ArArh. Celok 126: 283-289. Strunecka, A. (1999) Aluminum plus Fluoride: a new deadly duo. Dement. 1: 2- 3. Long, Y-G, Wang, Y-N, Chen, J., Jiang, S-F, Nordberg, A., and Guan, Z-Z. (2002) Chronic fluoride toxicity decreases the number of acetylcholine receptors in the rat brain. Neurotox. Terat, 23: 751-757. Burgstahler, A. W. (1966) Fluoridated water and Down’s syndrome. Long abstract of a report of the 21st Conference of the International Society for Brain Research, Budapest. Takahashi, K. (1998) Fluoride-linked Down syndrome births and their estimated occurrence due to water fluoridation. Fluoride, 31: 61-73. Juan Carlos Molina is the Director of the Ferryra Research Institute at the University of Cordoba, Argentina, as well as holding his distinguished professor position there. He also is a visiting research professor at Binghamton University.
1
Disclaimer: The material in this document represents my opinions, unless otherwise noted. The content may be copied in part or in full without permission when used in a not for profit format. When used for other purposes, the permission of the author is required. The document is not intended to provide medical advice but rather for the sharing of knowledge and opinions of the author. Decisions about health advice should be based on a personal one-on-one basis with an appropriate physician.
Robert L. Isaacson
Department of Psychology & Centre for Developmental
and Behavioral Neuroscience
Binghamton University
Binghamton, NY 13902-6000
Phone: 607 777 6764



 The two decay products of uranium:
The two decay products of uranium: More info → HERE ←
More info → HERE ← Yasser Arafat – 24 August 1929 – 11 November 2004
Yasser Arafat – 24 August 1929 – 11 November 2004