We at ‘Fluoridation Queensland’ do NOT support the addition
of any fluorides to: food, water, or air, this includes adding
it to salt, milk, baby formula and toothpaste. We are
not happy about its use in medicines, weapons,
insecticides, fungicides, anaesthetics,
and some industrial processes,
especially those that cause damage
to the earth’s atmosphere.
We have presented only a few papers from around the world.
Over 500 new papers on fluorides are added each year.
We suspect many more exist in the non-English press.

Dr. Burhenne ↓

We reserve the right to publish any of our outgoing correspondence
and incoming responses, or non-responses, on our
web sites as part of our policy of the-
❝ Citizen’s Right-To-Know ❞
The information on this website IS intended to help diagnose or treat disease.
It is provided for educational and informational purposes only.
Please consult an inappropriate professional
if you have any more concerns?
~ ‘A Wince of Dentists’ ~
·——«‹♦›»——·
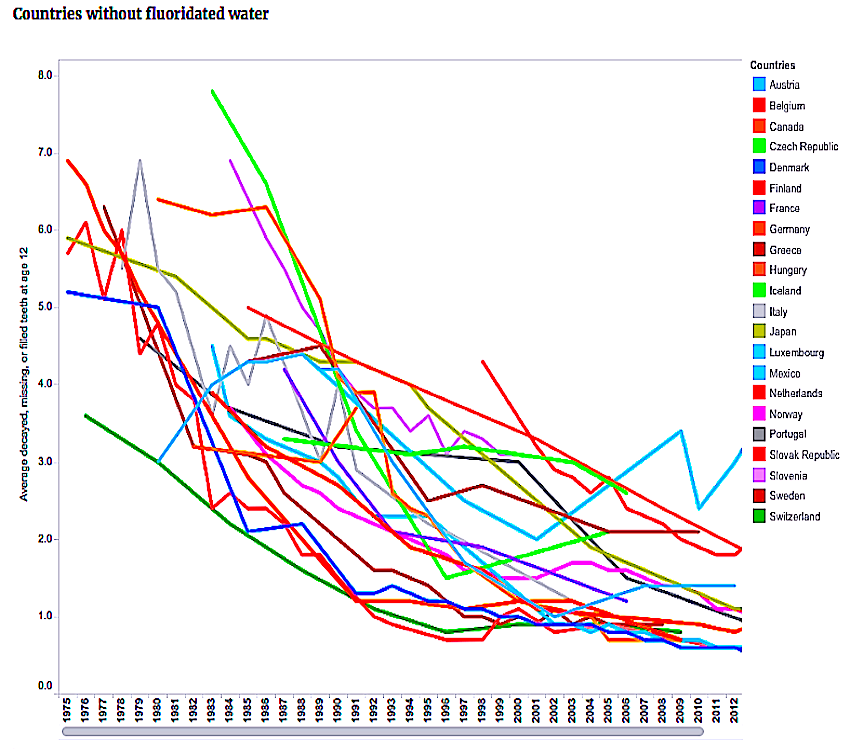
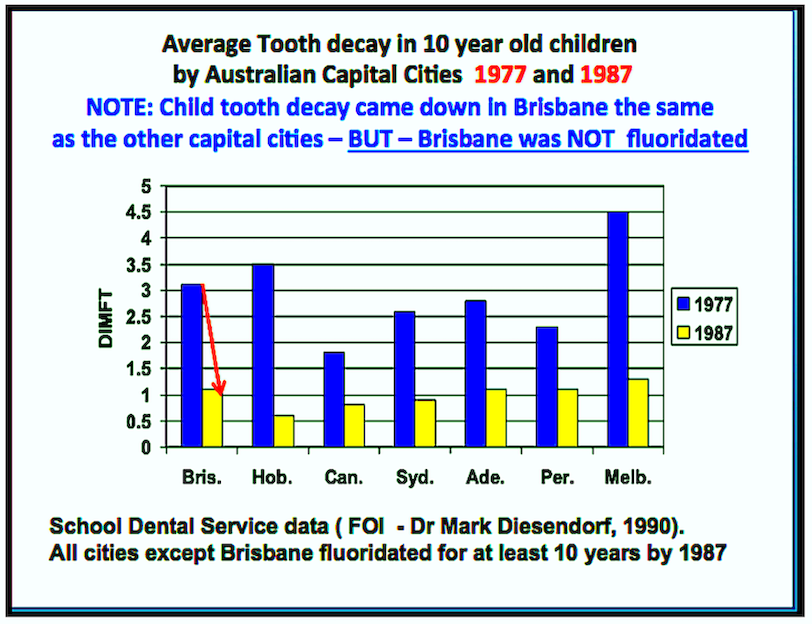
Decline Of Caries After The Cessation Of Fluoridation ↓

“The multi-billion dollar international conglomerates,
which benefit from tooth decay and fluoride sales,
pour money into organized dentistry, which is
behind virtually every fluoridation initiative.
Money makes dentistry politically powerful
and enables them to get laws passed to
benefit dentists’ bottom line.
However as evidenced by this and numerous other web sites,
dentists, cities, and nations are abandoning fluoridation and mercury
fillings. ‘Fluoridation Queensland’ will not rest until, in keeping with other
nations, mercury and fluorides are no longer used by Australian dentists.
·—————————–«‹♦›»—————————–·
·———————-«‹♦›»———————-·
The Fluoridation of Public Water Supplies Act (1963)
Why Not? – Timing, Circumstance,
A Review Of A Review
by Dr Harry Akers ↓
·————–«‹♦›»————–·
See also ↓
·———«‹♦›»———·

This work has been ignored and was out of print.
↓ but is now available see full text below ↓
Scientific Criticisms and Fluoride Dangers – by Philip R.N.Sutton D.D.Sc.
(Melb.), L.D.S., F.R.A.C.D.S. Formerly Senior Lecturer in Dental Science,
Senior Research Fellow

Fluoride The Aging Factor
THE ABSURDITIES OF WATER FLUORIDATION:
Scientists, dentists and professionals (even cooks) have been
persecuted, censored, or harassed when they oppose
the ‘Cult of Water Fluoridation’.

‘Second Look’ ↓ ↓ ↓

RUDOLF ZIEGELBECKER – Graz Austria:
❝ … So while fluoridation is neither effective nor safe, it continues to provide a convenient cover for many of the [vested] interests who stand to profit from the public being misinformed about fluoride. Unfortunately, because [some] government officials have put so much of their credibility on the line defending fluoridation, it will be very difficult for them to speak honestly and openly about this issue.
 As with the case of mercury amalgams, it is difficult for institutions such as the
As with the case of mercury amalgams, it is difficult for institutions such as the
American Dental Association to concede health risks because of the liabilities
…….waiting in the wings if they were to do so.




❝ The illiterate of the 21st century will not be those who can’t read or write –
but those who cannot learn, unlearn, and relearn. ❞
( May be he had a bad dentist! )

His work is important for reasons beyond its specific scientific findings.
His work was motivated by the assumption that ingested fluoride was beneficial!





Dentists Are Doing Very Well, Thank You – Despite Fluoridation.
Fluoridation hasn’t hurt dentistry’s bottom line at all.
In fact, dentistry is big business today despite 7 decades of water fluoridation and a glut of fluoridated dental products.
Americans spent about $108 billion on dentists in 2011, an inflation-adjusted increase from $64 billion in 1996, according to the General Accounting Office. But a dental crisis still exists.
Since fluoridation doesn’t reduce tooth decay, whose best interests are served
by protecting fluoride’s image?
The Global Toothpaste Market is expected to reach $12.6 billion by 2015,
according Global Industry Analysts, Inc.

[ A good reason to drink plenty of water! ]



Original full text → HERE

– FLUORIDATION – THE PROFITABLE LIE –
THE WALL STREET JOURNAL SAYS:
❝ Dentists Are Big Political Players and do all they can to preserve their monopoly.”
— Huge donations secure their political agendas:
They boasted the largest single health-care PAC in 2008, gave nearly $13 million to state and local politicians in 2010, raising the question: What do dentists want? Alicia Mundy has details on
‘The News Hub’.

Dental Crisis in America: –
After over 72 years of water fluoridation foisted upon Americans by the American Dental Association and its constituents groups, a Dental Crisis exists in America.
Senator Sanders introduced legislation to remedy this situation which is endorsed by 37 groups but not the ADA because it includes funding for Dental Therapists which would infringe upon dentists lucrative monopoly. The ADA prefers fluoridation because it doesn’t stop tooth decay and doesn’t hurt their bottom line. In fact, dentists are making lots of money covering up fluoride-stained teeth with expensive veneers because American children are now over-fluoridated with up to 60% affected with dental fluorosis – white spotted, yellow, brown and/or pitted teeth. Veneers cost about $1,000 a tooth.
For some Americans, dental care means a sturdy chair, a fluoride swish, and a free toothbrush.
But for one in three Americans, it’s a nightmare, including astronomical bills, crippling credit card debt, panicked visits to an emergency room, and life-threatening disease. . . .


Flagitious Lallation


Dr Jackie Robinson,
Associate Director, Continuing Education in Dentistry,
University of Sydney
[ Follow the money! ]


….→ Fluoridation on trial – 21 May 2018
Largely unseen, a lawsuit that aims to outlaw public water
fluoridation is moving forward in the federal District Court of Northern California

Delayed Tooth Eruption
This delay, at least partly, could be due to the teeth of children in fluoridated areas
erupting (breaking through the gums) at a slightly older age, and
therefore being exposed to decay-producing
factors for a shorter period:

Physical chemical effects of zinc on in vitro enamel…
“…Dean and his coworkers, although apparently convinced that the higher fluorine content of the waters of the former cities accounted for the lower incidence of dental caries, were somewhat hesitant to conclude that the fluorine content of the water was the only factor involved because of other differences in the mineral composition of the water supplies, primarily in the amount of calcium and magnesium…” –
Margaret Cammack Smith – (1935)



COMPETENCY CHECK: Airline pilots are required to undertake regular proficiency
checks to ensure they continue to be competent and aware of new regulations.
It would seem that the dental and medical professions are lax on this:
– They still promote fluoridation.
.


[ She is very unhappy about our web sites! ]
… A study of dentist and dental assistants in the Netherlands
found 50% higher rates of spontaneous abortions, stillbirths,
and congenital defects than for the control group,
with unusually high occurrence of spina bifida.
Full original text → HERE
·——«‹◊›»——-




The complete book with photos ↓ ↓ ↓
Fluorine Intoxication by Danish researcher Kaj Roholm. Published 1937, Copenhagen. Scientific research on the toxic effects of fluoride exposure and fluoride intoxication via ingestion. Studies on humans, plants and animals. Thirty chapters with a forty-page bibliography. Black and white plates of visible effects of fluoride exposure included…
Fluoride intoxication: a clinical-hygienic study with a review of the literature and some experimental investigations. London: H.K. Lewis Ltd.
“The teeth may be delayed in eruption and have abnormalities in size, shape and position, and resistance of such teeth is low.” p. 272
“The pathological enamel is brittle and readily chips off. The incisors wear down abnormally, which sometimes leads to defective occlusion and, as a secondary phenomenon, abnormal growth of opposing teeth. The incisors often exhibit lateral deviations; growth is retarded.” p. 272
The above research is
STILL VALID AFTER 78 YEARS
Come on ADA, it must be time to catch up!



EXTRACT FROM THE AUSTRALIAN DENTAL ASSOCIATION’S
WEB SITE DISCLAIMER:
❝ … The ADA Inc. shall not be responsible for information
provided herein under any theory of liability or indemnity.
Liability of ADA Inc., if any, for damages (including, without
limitation, liability arising out of contract, negligence,
strict liability, tort or patent or copyright infringement)
shall not exceed the fees paid by the user for the
particular information or service provided … ❞
( Sounds very mumblecrust – but it fills us all with confidence ! )



LARGE FILE → HERE ←


ADA / Qld. Labour Party Collusion :
The Queensland Health Department, funded
The Australian Dental Association Queensland Branch,
$220,000 as a CONtribution
to its pro-fluoridation campaign.
The request for this funding was directed to
the Hon. Stephen Robertson MP.
The Minister for Health at the time – Feb. 2006.


A Bibliography of Scientific Literature on Fluoride



FLAGITIOUS LALLATION

Jason M. Armfield
(Member of IADR)
❝ … Statements regarding the scientific controversy surrounding water fluoridation are generally regarded as artefacts of anti-fluoridationist activity, with actual scientific debate over water fluoridation being resolved decades ago. Almost all major dental and health organisations either support water fluoridation or have found no association between it and adverse health effects [20]. Nonetheless, propagating the idea of an ongoing scientific debate gives the illusion of scientific uncertainty and is a favoured tactic of water fluoridation opponents. In 1978, Consumer Reports published a two-part series on fluoridation that concluded:
The simple truth is that there’s no “scientific controversy” over the safety of fluoridation. The practice is safe, economical, and beneficial. The survival of this fake controversy represents, in Consumers Union’s opinion, one of the major triumphs of quackery over science in our generation.” [21] And yet, more than a quarter of a century after these words were printed the manufactured ‘controversy‘
shows no signs of diminishing. – [ Oh what a surprise! ]
The review estimated the prevalence of fluorosis (mottled teeth) and fluorosis of aesthetic concern at around 48% and 12.5% when the fluoride concentration was 1.0 part per million, 9 although the quality of the studies was low. The evidence was of insufficient quality to allow confident statements about other potential harms (such as cancer and bone fracture). The amount and quality of the available data on side effects were insufficient to rule out all but the biggest effects
Well said Mr. Armfield,
however, history has shown us that you have not been
keeping up with the recent science from overseas.
We hope our web site will help to keep you updated…
Extract: from letter by Dr. Jason Armfield
Australian Research Centre For Population Oral Health
University Of Adelaide:
ARCPOH Response to USHHS
(United States Health Service)
❝… Hence, fluoride exposures of importance to dental fluorosis occur across the early childhood years. Fluoride exposures of importance to dental caries occur across the whole life-course. Developing policy to influence those exposures needs to take account of this fundamental difference. Actions to reduce fluorosis should be targeted at fluoride exposures that affect children in the early childhood years, while maintaining fluoride exposures across life for the prevention of dental caries… ❞
Our Comments:
Well, what you are saying here,- in a round about way,
or are trying to cover up, is that dental fluorosis only
occurs during tooth formation, so reduce fluoride
from all other sources until after teeth have erupted, and no
NO HARM WILL BE VISIBLE, but do not lower the dose
in drinking water. However the evidence is, that damage from
fluoridation continues to the rest of the body after the teeth
are formed, but will not be recorded on the teeth –.
We do not think your response to USHHS is ethical!
Please see the research papers on this web site, or our links
to them, including animal studies, which support our comments.
Yours truly,
‘Fluoridation Queensland’

Artificial Water Fluoridation – No Benefit – Definite Harm
National data set collected in the U.S. in 1986-1987 (more than 16,000 children, ages 7-17, ↓

MORE OF OUR CONCERNS:
Caries / Dental Fluorosis
Because dental fluorosis only occurs during tooth formation, fluoride levels in water and exposure to fluoridated toothpaste needs to be low in children, or there will be evidence of harm.
Skeletal fluorosis can be misdiagnosed as arthritis,
and dental fluorosis would reveal the real problem…
Consumption of fluoridated water is difficult to monitor:
Fluid intake is relative to the “ambient air temperature”, work,
sport and exercise regimes. The availability of ‘clean’
bottled water, and RO filters, which are now commonplace
in many Australian households, and will distort the [your] data.
Because fluoride is medication, the dilemma will always be the dose, — 1ppm is a rate – not a dose,
and is not related to body weight, or many other variables
including nutritional status, kidney function, weather, climate,
latitude, elevation and daily temperature variations at the fluoridation plant.
Medication by thirst, is scientifically irresponsible.
The observation of harm with fluoridation is modified by cross contamination,- some foods and drinks will be crossing borders of reticulated water supplies, some being fluoridated and some not, or at different levels (ppm).
No amount of long-winded circumlocution, technical language or
extensive discussions of the “appropriateness of study design “
can disguise the simple and obvious problems of trying to deliver medication via the public water supply at a defined dose… it defies logic, is contemptuous of science and it is professionally irresponsible to pretend otherwise…

There are only preliminary estimates available regarding how much F. is absorbed through our skin and lungs when we stand naked in our shower or soak in the bath contaminated by fluoridated water – and we also pay for this indignity in our water bill.

Cognitive Vulnerability:
A MODEL OF THE ETIOLOGY OF FEAR
By Jason M. Armfield
“ Fear is a powerful and considerably aversive human emotion.”
… We agree with the above statement. Your paper is interesting and is widely researched but makes no mention of nutrition? We feel this is an major oversight. Animal and human behavior, reproduction, health and mental state are dependent on balanced nutrition and freedom from mind-altering chemicals and heavy metals.
Deficiencies of zinc, (especially for men and boys), some of the B vitamins, and vitamin K2 are well established and documented as essentials for a healthy body and a stable personality.
Excess sugar, aspartame and alcohol can cause mental disturbances indeed any chemical that can interfere with the pineal gland will have psychological impacts. Bad parenting is a significant factor, but the Hopewood experience provided evidence that even this can be over-come with a good diet and environment. Gov.info.
Fluoride in its various forms interferes with many nutrients, enzymes and minerals and the pineal gland, and as such is contraindicated. This last statement however may be at odds with your training and profession, but science is science and it is often in a lifetime that at least one change of belief is required in the bright dawn of a new revelation. Research papers from the 1930s and recent times, especially recent research from countries not influenced by U.S.A, and its commercial interests, confirm and reaffirm that fluoride in any of its forms is a hazard to plants, animals, enzymes, and humans.
Dr. Jason M Armfield:
❝ …. Any action to lower the fluoride levels must be carefully
considered in order not to undermine the protective effect *…. ❞
[* read – Protection of ‘Our Water Fluoridation Policy’ ]
A recent scientific report shows that fluoride disturbs the enzymes casein kinase II och alkaline phosfatase, which causes a disordered enamel formation. Thus, what dentists call innocent stains on the teeth is actually a symptom of chronic fluoride poisoning in the infant. Enamel damaged by fluorosis has been shown more susceptible to caries than normal enamel, recently in study of native children in South Africa who never experience any type of dental care. When considering the biological nature of fluorosis, it is not hard to understand that children during the period of dental development should avoid fluoride as much as possible, i.e. they should not have fluoride tablets, fluoridated chewing gum or drink fluoridated water or consume fluoridated salt or fluoridated milk.
Full text → HERE
Your internet article listed below hints at desperation,
and is almost a promotion of our cause. Your awareness
of the dangers of fluoridation and fluoride contamination
suggests that mammon is more influential than true science.
“methinks thou dost protest too much“
‘When public action undermines public health:
a critical examination of anti-fluoridationist literature’.
Fluoridation is NOT controversial. It is medication.




❝ There is no right way to do the wrong thing.” – Oren Arnold

When the debate is lost, slander becomes the tool of the loser. – Socrates


European Journal Of General Dentistry – 2013
Is fluoride still a pivot of preventive dentistry?
Authors: Poonam Mahajan, KL Veersha, Ajay Mahajan
Pages: 20 – 24
Abstract:
Poonam Mahajan, KL Veersha, Ajay Mahajan
European Journal of General Dentistry 2013 2(1):20-24
Fluoride is considered the corner stone of the preventive dentistry. Fluoride has both beneficial and detrimental effects on human health.
In terms of dental health, the prevalence of dental caries is inversely related to the concentration of fluoride in drinking water; while there is a dose-response relationship between the concentration of fluoride in drinking water and the prevalence of dental fluorosis.
Fluoride has a statistically significant association with a wide range of adverse effects like increased risk of bone fractures, decreased thyroid function, and lowered intelligent quotient, arthritic-like condition, early puberty and possibly, osteosarcoma. The aim of the present review is to discuss the current status of fluorides in dentistry in view of its benefits and adverse effects.
Citation: European Journal of General Dentistry 2013 2(1):20-24
PubDate: Sat,2 Feb 2013
DOI: 10.4103/2278-9626.106797
Issue No: Vol. 2, No. 1 (2013)




Professor Niyi Awofeso
School of Population Health
… Awofeso’s analysis reinforces Shaw’s conclusion by showing
that most of the arguments used to vindicate fluoridation
do not stand up to careful ethical scrutiny…




↑ THIS IS A VERY LONG LIST ↑

Bite Magazine and website:

A new study at the University of Sydney will try to find out what’s going wrong
with their teeth. The tiny teeth of Australian toddlers are rotting and dental
researchers at the University of Sydney are poised to start a long-term
study to find out why.
Our comment:
It looks like artificial water fluoridation is still not working.
Fluoride does not work for sheep, see below ↓
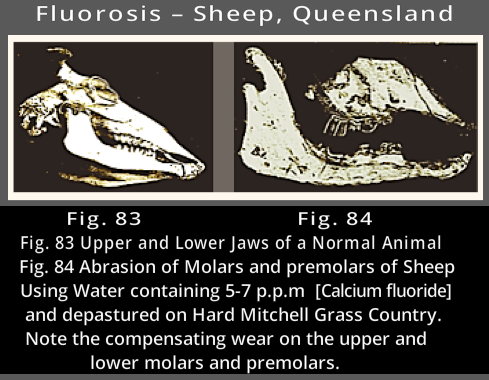
See more photos like this from ↓

See also → Fluoride ← Credit Dr. Mercola

…This apparatus
has been erected
to lie and obfuscate this issue. It cannot
tolerate a single chink in its armour of deceit.
No compromise, no partial admission is possible without
the integrity of the whole edifice of deception being threatened.
❝ It is difficult to get a man to understand something
when his salary depends upon his not understanding it. ❞
— Upton Sinclair —
USA To Lower Fluoride Levels: ATLANTA 7 January 2011 (extracts from reports) In a remarkable turnabout, federal health officials say many Americans are getting too much fluoride, and it’s causing splotches on children’s teeth and perhaps other, more serious problems.
The U.S. Department of Health and Human Services announced plans Friday (7 Jan) to lower the recommended level of fluoride in drinking water for the first time in nearly 50 years, based on a fresh review of the science. The announcement is likely to renew the battle over fluoridation, even though the addition of fluoride to drinking water is considered one of the greatest public health successes of the 20th century. One reason behind the change: About 2 out of 5 adolescents have tooth streaking or spottiness because of too much fluoride, a government study found recently. In extreme cases, teeth can be pitted by the mineral — though many cases are so mild only dentists notice it. The problem is generally considered cosmetic and not a reason for serious concern.
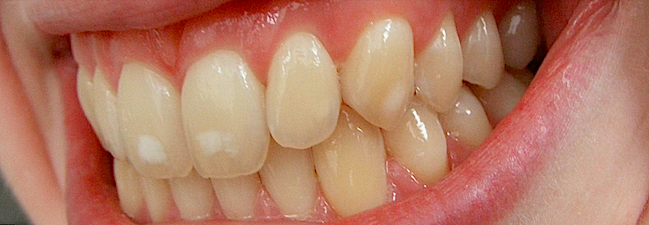
The splotchy tooth condition, fluorosis, is unexpectedly common in youngsters ages 12 through 15 and appears to have grown more common since the 1980s, according to the Centers for Disease Control and Prevention. But there are also growing worries about more serious dangers from fluoride.
The Environmental Protection Agency released two new reviews of research on fluoride Friday.
One of the studies found that prolonged, high intake of fluoride can increase the risk of brittle bones, fractures and crippling bone abnormalities. Critics of fluoridated water seized on the proposed change [on] Friday to renew their attacks on it — a battle that dates back to at least the Cold War 1950s, when it was denounced by some as a step toward Communism. Many activists nowadays don’t think fluoride is essential, and they have praised the government’s new steps.

Extract from:
FLUORIDE IN DRINKING WATER
A SCIENTIFIC REVIEW OF EPA’S STANDARDS
Committee on Fluoride in Drinking Water
Board on Environmental Studies and Toxicology
Division on Earth and Life Studies
Credit to:
NATIONAL RESEARCH COUNCIL OF THE NATIONAL ACADEMIESTHE NATIONAL ACADEMIES PRESS
Washington, D.C.
Available Online → HERE ←
ENAMEL FLUOROSIS
❝ Fluoride has a great affinity for the developing enamel because tooth apatite crystals have the capacity to bind and integrate fluoride ion into the crystal lattice (Robinson et al. 1996). Excessive intake of fluoride during enamel development can lead to enamel fluorosis, a condition of the dental hard tissues in which the enamel covering of the teeth fails to crystallize properly, leading to defects that range from barely discernable markings to brown stains and surface pitting. This section provides an overview of the clinical and histopathological manifestations of enamel fluorosis, diagnostic issues, indexes used to characterize the condition, and possible mechanisms.
CLINICAL AND HISTOLOGICAL FEATURES
Enamel fluorosis is a mottling of the tooth surface that is attributed to fluoride exposure during tooth formation. The process of enamel maturation consists of an increase in mineralization within the developing tooth and concurrent loss of early-secreted matrix proteins. Exposure to fluoride during maturation causes a dose-related disruption of enamel mineralization resulting in widening gaps in its crystalline structure, excessive retention of enamel proteins, and increased porosity. These effects are thought to be due to fluoride’s effect on the breakdown rates of matrix proteins and on the rate at which the by-products from that degradation are withdrawn from the maturing enamel (Aoba and Fejerskov 2002).
Clinically, mild forms of enamel fluorosis are evidenced by white horizontal striations on the tooth surface or opaque patches, usually located on the incisal edges of anterior teeth or cusp tips of posterior teeth. Opaque areas are visible in tangential reflected light but not in normal light. These lesions appear histopathologically as hypomineralization of the subsurface covered by a well-mineralized outer enamel surface (Thylstrup and Fejerskov 1978). In mild fluorosis, the enamel is usually smooth to the point of an explorer, but not in moderate and severe cases of the condition (Newbrun 1986). In moderate to severe forms of fluorosis, porosity increases and lesions extend toward the inner enamel. After the tooth erupts, its porous areas may flake off, leaving enamel defects where debris and bacteria can be trapped. The opaque areas can become stained yellow to brown, with more severe structural damage possible, primarily in the form of pitting of the tooth surface.
Enamel in the transitional or early maturation stage of development is the most susceptible to fluorosis (Den Besten and Thariani 1992). For most children, the first 6 to 8 years of life appear to be the critical period of risk. In the Ikeno district of Japan, where a water supply containing fluoride at 7.8 mg/L was inadvertently used for 12 years, no enamel fluorosis was seen in any child who was age 7 years or older at the start of this period or younger than 11 months old at the end of it (Ishii and Suckling 1991). For anterior teeth, which are of the most aesthetic concern, the risk period appears to be the first 3 years of life (Evans and Stamm 1991; Ishii and Suckling 1991; Levy et al. 2002a). Although it is possible for enamel fluorosis to occur when teeth are exposed during enamel maturation alone, it is unclear whether it will occur if fluoride exposure takes place only at the stage of enamel-matrix secretion. Fejerskov et al. (1994) noted that fluoride uptake into mature enamel is possible only as a result of concomitant enamel dissolution, such as caries development. Because the severity of fluorosis is related to the duration, timing, and dose of fluoride intake, cumulative exposure during the entire maturation stage, not merely during critical periods of certain types of tooth development, is probably the most important exposure measure to consider when assessing the risk of fluorosis (Den Besten 1999).
MECHANISMS
Dental enamel is formed by matrix-mediated biomineralization. Crystallites of hydroxyapatite (Ca10(PO4)6(OH)2) form a complex protein matrix that serves as a nucleation site (Newbrun 1986). The matrix consists primarily of amelogenin, proteins synthesized by secretory ameloblasts that have a functional role in establishing and maintaining the spacing between enamel crystallites. Full mineralization of enamel occurs when amelogenin fragments are removed from the extracellular space. The improper mineralization that occurs with enamel fluorosis is thought to be due to inhibition of the matrix proteinases responsible for removing amelogenin fragments. The delay in removal impairs crystal growth and makes the enamel more porous (Bronckers et al. 2002). DenBesten et al. (2002) showed that rats exposed to fluoride in drinking water at 50 or 100 mg/L had lower total proteinase activity per unit of protein than control rats. Fluoride apparently interferes with protease activities by decreasing free Ca2+ concentrations in the mineralizing milieu (Aoba and Fejerskov 2002).
Matsuo et al. (1998) investigated the mechanism of enamel fluorosis in rats administered sodium fluoride (NaF) at 20 mg/kg by subcutaneous injections for 4 days or at 240 mg/L in drinking water for 4 weeks. They found that fluoride alters intracellular transport in the secretory ameloblasts and suggested that G proteins play a role in the transport disturbance. They found different immunoblotting-and-pertussis-toxin-sensitive G proteins on the rough endoplasmic reticulum and Golgi membranes of the germ cells of rats’ incisor teeth.
HEALTH ISSUES AND CLINICAL TREATMENT
Whether to consider enamel fluorosis, particularly the moderate to severe forms, an adverse cosmetic effect or an adverse health effect has been the subject of debate for decades. Some early literature suggests that the clinical course of caries could be compromised by untreated severe enamel fluorosis. Smith and Smith (1940, pp.1050-1051) observed,
“There is ample evidence that mottled teeth, though they be somewhat more resistant to the onset of decay, are structurally weak, and that unfortunately when decay does set in, the result is often disastrous.”
Caries once started evidently spreads rapidly. Steps taken to repair the cavities in many cases were unsuccessful, the tooth breaking away when attempts were made to anchor the fillings, so that extraction was the only course.” Gruebbel (1952, p.153) expressed a similar viewpoint: “Severe mottling is as destructive to teeth as is dental caries. Therefore, when the concentration is excessive, defluorination or a new water supply should be recommended. The need for removing excessive amounts of fluorides calls attention to the peculiar situation in public health practice in which a chemical substance is added to water in some localities to prevent a disease and the same chemical substance is removed in other localities to prevent another disease.” Dean advised that when the average child in a community has mild fluorosis (0.6 on his scale, described in the next section), “… it begins to constitute a public health problem warranting increasing consideration” (Dean 1942, p. 29).
There appears to be general acceptance in today’s dental literature that enamel fluorosis is a toxic effect of fluoride intake that, in its severest forms, can produce adverse effects on dental health, such as tooth function and caries experience. For example:
-
“The most severe forms of fluorosis manifest as heavily stained, pitted, and friable enamel that can result in loss of dental function” (Burt and Eklund 1999).
-
“In more severely fluorosed teeth, the enamel is pitted and discolored and is prone to fracture and wear”
(ATSDR 2003, p. 19).
-
“The degree of porosity (hypermineralization) of such teeth results in a diminished physical strength of the enamel, and parts of the superficial enamel may break away … In the most severe forms of dental fluorosis, the extent and degree of porosity within the enamel are so severe that most of the outermost enamel will be chipped off immediately following eruption” (Fejerskov et al. 1990, p. 694).
-
“With increasing severity, the subsurface enamel all along the tooth becomes increasingly porous … the more severe forms are subject to extensive mechanical breakdown of the surface” (Aoba and Fejerskov 2002, p. 159).
-
“With more severe forms of fluorosis, caries risk increases because of pitting and loss of the outer enamel” (Levy 2003, p. 286).
-
“ … the most severe forms of dental fluorosis might be more than a cosmetic defect if enough fluorotic enamel is fractured and lost to cause pain, adversely affect food choices, compromise chewing efficiency, and require complex dental treatment” (NRC 1993, p. 48).
Severe enamel fluorosis is treated to prevent further enamel loss and to address the cosmetic appearance of teeth. Treatments include bleaching, microabrasion, and the application of veneers or crowns. Bleaching and microabrasion are typically used with the mild to moderate forms of enamel fluorosis. Bleaching is the least invasive procedure, but does not eliminate the dark stains associated with severe enamel fluorosis. Microabrasion involves the controlled abrasion of enamel to remove superficial stains… ❞
OUR NOTE: The full text is available on line, → HERE ←
However it does not address the matter of the nutrition, and
the variations in results e.g. vitamin C, magnesium, intake etc.
~ Don’t Let Your Diet Be Dictated By Corporate Agendas ~

❝ …fluoride reduces the level of thyroid hormone during tooth development, by activation calcium-transducing G-protein receptor G q/11, there is delayed tooth eruption, delayed removal of enamel matrix proteins, and delayed enamel maturation.
The evidence of the deficiency is seen later with mottled teeth… ❞
Extract from:
by Bruce Spittle.
The complexities of halogen chemistry are legion and it is that very complexity that is employed by the less scrupulous fluorine advocates to stifle protest from their adversaries with pseudo- scientific, convoluted terminology, which very often they themselves do not understand.
Very much a matter of ‘the blind leading the blind’.
Fluorine is an element. Atomic number 9. Atomic weight 18.9984. Symbol F. Fluorine is a halogen. There are five halogens known to contemporary science. In alphabetical order these are:
Astatine, Bromine, Chlorine, FLUORINE and Iodine.
Fluorine is one of the most poisonous substances known to the science of toxicology. The word ‘halogen’ is derived from the Greek, hals meaning salt and genes meaning born. The word refers to the propensity of the element to combine with Sodium.
Examples are: Sodium Bromide, Sodium Chloride, Sodium Fluoride and so forth.
This ‘mixture’ of a halogen and one or more other substances is called a ‘halide’, the letter “IDE” being affixed to the first letters of the particular halogen title. Thus Bromine compounded with something else becomes a Bromide. A Chlorine compound likewise becomes a Chloride and a Fluorine compound becomes a Fluoride.
The other ingredient(s) of the halide are included in the title to completely identify the halide chemically. So Sodium mixed with Chlorine becomes Sodium Chloride. Potassium mixed with Bromine becomes Potassium Bromide and Calcium mixed with Fluorine becomes Calcium Fluoride and so on for literally thousands of simple and complex combinations.
For all practical purposes the element FLUORINE cannot exist as a single entity. It must always associate with another substance or substances to exist.
That is to say it is only found as a fluoride.
In this need for existence, Fluorine, more so than the other halogen elements, will combine avidly with almost any other substance or substances, taking its deadly potential along with it to impart a lethal hazard to what was formerly a most innocent and benign material. The resultant degree of toxicity is not in debate here.
A well recognized example of this ‘chemical corruption’ is in the medicinal drug “Fluorouracil” whereby the vital and totally inoffensive human metabolite “uracil” is converted into a deadly ‘cytotoxic’ (meaning – cell destructive) killer compound.
So then simple (duplex) Fluorine based compounds can combine or be combined with one or more other virtually harmless substances to create a chemical killer of considerable complexity and extreme danger. A ‘triplex’ Fluoride is Sodium Fluoroacetate, marketed under the cover name of Compound 1080.
Another is Fluoroacetamide (Compound 1081).
The designed deadliness of both these FLUORIDE products is legend and promoted under the 1080 and 1081 labels but the close chemical relationship with the Sodium Fluoride in your water supply is vehemently denied. Much less admitted.
Other examples of multiple fluorides are “SARIN”
(Isopropyl methyl-phospho FLUORIDATE) and
“SOMAN” (Pinacolyl methylphos-phonoFLUORIDATE).
Both these ‘fluorides’ are nerve gasses, one drop of either of which,
absorbed through the skin, will kill an adult human, in seconds.
In the Martindale Extra Pharmacopoeia, under the heading
Sodium Fluoride is the sub-heading “and other Fluorine Compounds”
which include both 1080 and 1081.
Other data and relevant physiologic aspects of Fluorides have been set out in
“The Dickinson Statement”. ( See also 2013 Expanded Edition)
In closing this paper it should be pointed out that Fluorides are toxic to animals in a number
of chemical and bio-chemical ways.
CORROSIVE, ANTI-METABOLIC, ALLERGENIC, GENETIC, CARCINOGENIC, SYSTEMIC, ENZYME INHIBITING and CUMULATIVE are among the popular description of the toxic activity of fluorides.
As an “acute” poison, a comparatively large dose may be needed to cause extinction but as a “chronic” toxin, neither the microscopic amounts needed nor the period of exposure (to cause death) have ever been evaluated. However, as an awesome indication, time lapse photography of the activity of living mammalian cells under the influence of
ONE PART OF SODIUM FLUORIDE TO 60 MILLION PARTS OF WATER
demonstrates almost immediate extinction of the cells.
Fluoridated tap water contains an “average” of –
TWO PARTS OF SODIUM FLUORIDE TO ONE MILLION PARTS OF WATER.
Suffice it now to say that in the final analysis all FLUORIDES are killers and who-so-ever administers, or causes to be administered any Fluoride on any pretext, by accident or by design to any person of any age is, by simple definition a KILLER.
Ian E. Stephens, 8 June 1988. [ For more info by same author See → HERE ]
Although 1 part fluoride ion in one million is argued to be ‘safe’ for human consumption, understand that one liter of water fluoridated with 1.0 ppm fluoride ion contains around 3.18 billion billion atoms of fluoride ion.
The toxic action of fluoride resides in the fact that fluoride ions act as enzymatic
poisons, inhibiting enzyme activity and, ultimately,
interrupting metabolic processes such as
glycolysis and synthesis of proteins.
Do you enjoy tales of intrigue, conspiracy and corruption? Then ‘The Fluoride Deception’ by Christopher Bryson won’t disappoint. It’s a very well written, meticulously researched expose´ of the history of fluoridation, which Dr Robert Carton (formerly of the US Environmental Protection Agency) has described as “the greatest case of scientific fraud of this (20th) century, if not of all time”. It’s a history of “greed, collusion, personal aggrandizement, corporate and government cover-up,” littered with human tragedy and the tattered careers of scientists who dared to try to expose the truth.
‘Esteemed Voices‘ have, for 50 years, warned the American public that water
fluoridation has dangerous long-term consequences to health:
“I am appalled at the prospect of using water as a vehicle for drugs. Fluoride is a corrosive poison that will produce serious effects on a long range basis. Any attempt to use water this way is deplorable.” Dr. Charles Gordon Heyd, Past President of the American Medical Association.


❝ Contrary to what is widely assumed, the toxicity of
fluoride is not always related to concentration.
Under certain conditions fluoride toxicity actually
increases as the concentration decreases.
This is what is known as a paradoxical effect. ❞

Letters sent “NO thanks” ↓
Pro-fluidationists are blinded by a diabolical ideology, which
does not allow them to draw rational conclusions.
This ideology is hostile the rights of individuals to choose
or reject their personal medical needs.
Iodine is a halogen. The halogens are a series of non-metal elemants:
Fluorine, F
Chlorine, Cl
Bromine, Br
Iodine, I
Astatine, At
The problem is that fluoride is a “bully.” Any time an atom of fluoride
and an atom of any other halogen are in the same vicinity, the
fluoride will displace the other halogen and take its place.
– Dr. Terry Tennant, MD, MD(H), PSc.,D

Editor’s comment:
This is universal, probably throughout time and most likely throughout
the cosmos. We realise that some dentists will see this as yet another
threat to their constant claims of fluoridation’s safety, but
to claim otherwise is to fly in the face of true science.

This one is interactive – Take a look ↑ ↑ ↑


” No physician in his right senses would prescribe for someone he has never met, whose medical history he does not know, a substance which is intended to create bodily change, with the advice: “Take as much as you like, but you will take it for the rest of your life because some people say that it can reduce tooth decay in children.” –
Dr. Peter Mansfield, Director Templegarth Trust, Birmingham, UK.

SALT FLUORIDATION is as much “forced medication” as water fluoridation.
“Universal” salt fluoridation – where all salt destined for human consumption is fluoridated – is worse than any water fluoridation measure, as no one can escape the measure. Salt has a quality worse than water: an individual’s preferred level of salt may be raised or lowered by progressively raising or lowering the actual level over a period of time. In other words, the amount of “salt sprinkled on” today might not be enough a month from now when just a little bit more will be added.







↓ Dental Fluorosis ↓
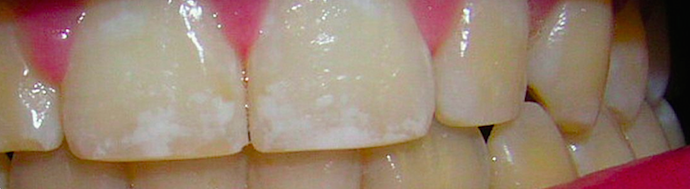

“ Fluoride-deficiency does not cause tooth decay.
Rotten diets cause rotten teeth and no
amount of fluoride changes that.“









 As with the case of mercury amalgams, it is difficult for institutions such as the
As with the case of mercury amalgams, it is difficult for institutions such as the![]()






















































